Accelerate your lab's
research today
Register for free — upgrade anytime.
Interested in getting a license? Contact Sales.
Sign up freeWritten by Keaun Amani
Published 2023-5-17
Macrocyclic peptides are a class of peptides characterized by a cyclic structure formed through the connection of the peptide chain's N- and C-termini. In other words, the amino acid sequence folds back on itself to create a ring structure. The ring is typically composed of 8 to 50 amino acids but can vary quite a bit.
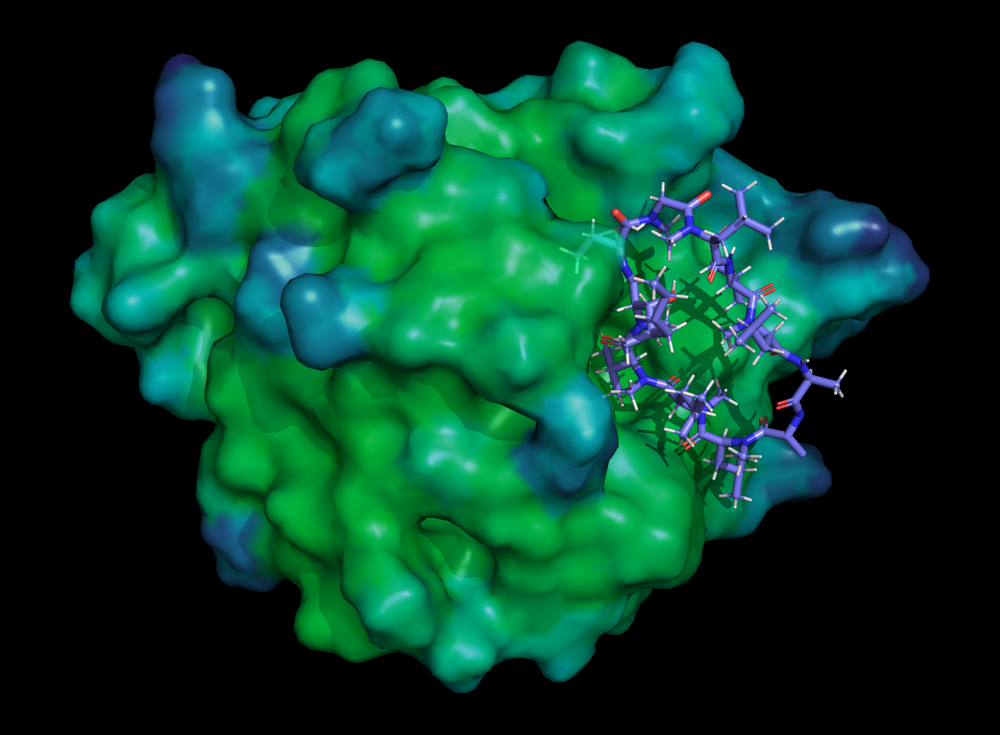
Macrocyclic peptides exhibit unique properties that make them attractive in various fields, including pharmaceuticals, biotechnology, and materials science. Some of the key characteristics and advantages of macrocyclic peptides include:
Enhanced stability: The cyclic structure provides increased resistance to enzymatic degradation, allowing macrocyclic peptides to have longer half-lives compared to linear peptides.
Improved target binding: The constrained and rigid structure of macrocyclic peptides allows for more precise and favorable interactions with biological targets, such as proteins or receptors. This enhanced binding affinity can lead to higher potency and selectivity.
Diverse target range: Macrocyclic peptides can target a wide range of biological molecules, including protein-protein interactions, enzymes, and receptors. They offer the potential to modulate complex biological pathways and can be engineered to bind specific targets with high affinity.
Oral bioavailability: The increased stability and target binding properties of macrocyclic peptides can facilitate their oral administration, allowing for more convenient delivery compared to injectable forms.
Structural diversity: Macrocyclic peptides can be chemically modified and diversified to improve their pharmacokinetic properties, stability, and selectivity. This versatility enables the development of libraries of macrocyclic peptides with varying activities for drug discovery and optimization.
Due to their favorable properties, macrocyclic peptides have gained significant attention in drug discovery efforts. Several macrocyclic peptide-based drugs have been developed or are under investigation for the treatment of various diseases, including cancer, infectious diseases, and metabolic disorders
Macrocyclic peptides are formed through a process called macrocyclization, which involves the formation of a covalent bond between the N- and C-termini of a peptide chain, creating a cyclic structure. This process can occur naturally within living organisms or be achieved synthetically in the laboratory.
Natural Formation: In nature, macrocyclic peptides are often synthesized by specialized enzymes called cyclases. These enzymes catalyze the formation of the peptide bond between the N- and C-termini of the peptide chain, effectively "stitching" them together. Cyclases typically recognize specific amino acid sequences and facilitate the cyclization process. The resulting macrocyclic peptide can exhibit enhanced stability and unique structural properties compared to linear peptides.
Synthetic Formation: Synthetic macrocyclic peptides can be created through various chemical methods. One common approach is solid-phase peptide synthesis (SPPS), which involves stepwise addition of protected amino acids onto a solid support. After elongating the peptide chain, a cyclization step is introduced. This can be achieved through different strategies, including:
These synthetic methods provide researchers with control over the size, structure, and properties of the macrocyclic peptides, allowing for the development of diverse applications in areas such as drug discovery, molecular recognition, and chemical biology.
AfCycDesign is a novel approach which leverages AlphaFold2 to predict and even design cyclic peptides. This technique essentially works by modifying the residue indices that the encode positional embeddings in AlphaFold2. By using an offset matrix the authors were able to generate cyclic peptides that were experimentally validated to form peptide bonds between the N & C termini.
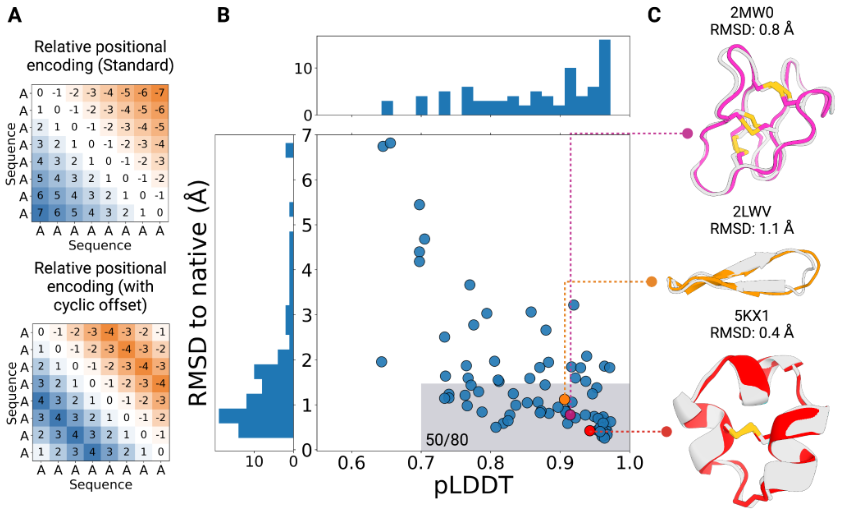
In addition, the authors state that AfCycDesign demonstrates a high level of accuracy in predicting the structures of native cyclic peptides from a single sequence. Out of 49 cases, 36 were predicted with strong confidence (pLDDT > 0.85) and exhibited a root mean squared deviation (RMSD) of less than 1.5 Å when compared to the native structure.
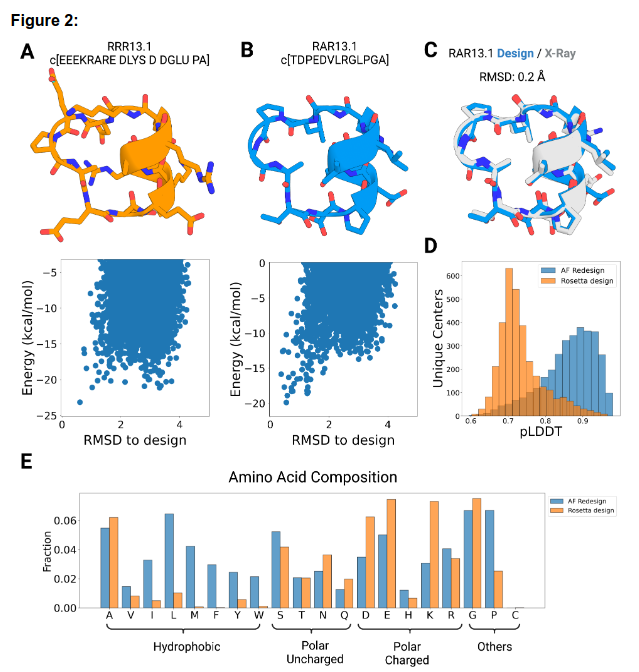
The authors also present computational techniques for designing sequences of peptide backbones generated by alternative backbone sampling methods, as well as for the creation of novel macrocyclic peptides.
Through extensive sampling, they explored the structural diversity of cyclic peptides spanning 7 to 13 amino acids. Their analysis identified approximately 10,000 distinct design candidates that were predicted to fold into the desired structures with a high level of confidence. Notably, when comparing seven sequences with varying sizes and structures to their corresponding X-ray crystal structures, they observed a close alignment (root mean squared deviation < 1.0 Å), underscoring the atomic precision achieved in their methodology.
The computational methods and scaffolds presented in this study serve as a foundation for tailoring peptides to specific therapeutic applications.
Neurosnap is a platform that provides easy access to tools and models like AfCycDesign and in this tutorial we're going to show you how to create your very own cyclic peptide using a template protein. The template protein refers to the input protein that you want to create a cyclic version of. In this case, we're going to use beta lactamase (1BTL) as the input protein and instruct the AfCycDesign service to try and create a cyclic protein with similar properties and structure to beta lactamase.
This can be done by heading over to our AfCycDesign service page and putting 1BTL within the Template Structure field. Next we need to specify the actual chain to use within the template structure. For 1BTL this is fairly straightforward as the structure only has one chain with the ID A which we then specify within the Chain field. After that we can submit the job and let Neurosnap handle the rest.
The results for this job are now available here. If you're interested in creating your own cyclic peptides create a Neurosnap account here!

In conclusion, AfCycDesign stands out as a remarkable application of AlphaFold2, leaving many individuals pleasantly surprised. As the field of cyclic peptides continues to gain prominence in research, the availability of powerful tools like AfCycDesign is expected to have a profound impact on accelerating advancements in this area. By leveraging this technology, researchers can unlock new possibilities and expedite their investigations in the dynamic realm of biochemistry.
By Amélie Lagacé-O'Connor
By Amélie Lagacé-O'Connor
By Danial Gharaie Amirabadi
By Danial Gharaie Amirabadi
By Danial Gharaie Amirabadi
By Danial Gharaie Amirabadi
Register for free — upgrade anytime.
Interested in getting a license? Contact Sales.
Sign up free